 
It is possible to quantify it as follows: It’s often regarded as a strong anesthetic for some surgical applications.ĬHCl3 has a molecular mass of 119.37 g/mol. It won’t allow the gas to escape from the bottle. During World War II, this gas was used to eliminate the people.Īs a result of this, chloroform(CHCl3) should be kept in a dark room with almost nil light penetration, tightly closed bottles up to the brim. It is known as carbonyl chloride(COCl2), also known as phosgene gas. In this blog from our previous post, we will look at the polarity of the CH2Cl2 molecule and find out if the molecule is polar or not.Ĭhloroform (CHCl3) is a highly flammable substance that reacts with light and air to produce toxic gas. It is important to decide whether the CHCl3 molecule is polar because polarity influences other physical properties. This occurs when the electronegativity value of the atoms in the CHCl3 molecule varies significantly. Polarity is a property that is found in CHCl3 molecule that has positive and negative charges separated in them. Physical properties of Trichloromethane(CHCl3).How to check the polarity of the CHCl3 molecule?. 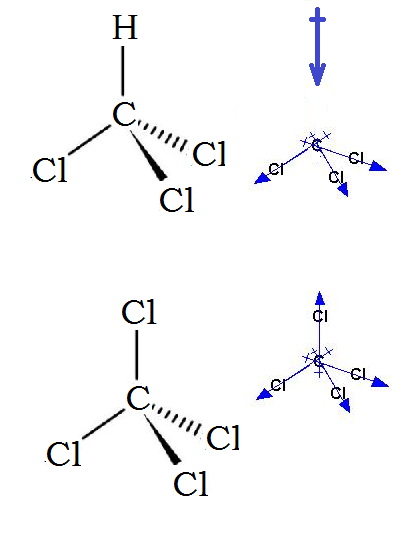
CHCl3 Lewis Structure, molecular geometry, and polarity:.Chlorine atoms are more electronegativity value than carbon and hydrogen atoms, and they are located at three of the pyramid’s vertices, pulling the negative charge in their direction, resulting in a polar molecule with a downward dipole. Is CHCl3 polar or nonpolar molecule, then? Because of its tetrahedral molecular geometry structure and the difference in electronegativity value of carbon(C), hydrogen(H), and Chlorine(Cl), CHCl3 is polar. It gives a sweet pleasant aroma and vaporizes very easily. CHCl3 is not miscible with water and creates a separate organic interracial layer. In this blog post, we try to answer all the above said questions in a detailed manner. The student used to ask ” is ChCl3 polar or nonpolar?”, “CHCl3 Lewis structure”, “CHCl3 molecular geometry”, and “polarity of CHCl3”. This molecule has three C-Cl bonds, Cl atom is having a higher electronegativity value. Methyl trichloride or trichloromethane is falling in the same family with the chemical formula of CHCl3. More the electronegativity of the atom, more is the tendency of the atom to attract the shared pair of electrons towards itself and more is the polarity of the molecule and vice-versa.Chlorinated hydrocarbons are generally used as a solvent in the chemistry laboratory. Note: The polarity of the molecule depends on the presence of the electronegative atom in the molecule. assertion is correct but reason is not correct is the correct answer. Reason: As discussed above, the Lewis dot is the structural representation of the valence electrons and it is not essential that the polar molecules should have at least one lone pair of electrons. So, methyl chloride is a polar molecule and thus, the assertion is correct. carbon, hydrogen and chlorine have different electronegativities and have symmetrical structure and the bond dipoles do not cancel each other. On the other hand, the Lewis dot diagram tells us about the number of the valence electrons present in an atom and helps to know whether the valence electrons exist as lone pairs of electrons or are present in the bond pair.Īssertion: In the methyl chloride, all the atoms i.e. 
On the basis of the bonds which show the net dipole in a molecule, we can check whether the given molecule is polar or nonpolar. the positive and the negative charges and the molecule have net dipole. By the polar molecule, we mean that molecule in which there is separation of the charges i.e. Now you can easily answer the statement.įirst of let’s discuss what is a polar molecule. Hint Polar molecules are those whose net dipole moment of the bond pairs do not cancel each other and have some net dipole moment and Lewis dot structure tells us about the number of valence electrons in an atom. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
